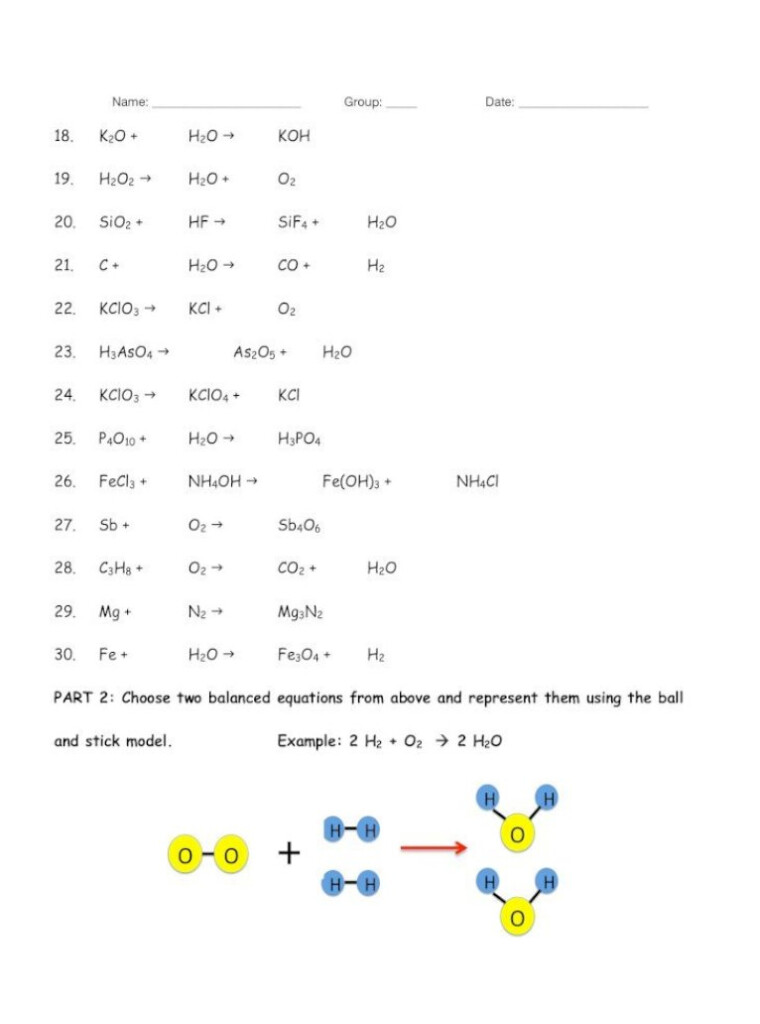
Chemistry Worksheet Balancing Equations Part 2 – This Chemistry Equations Worksheet will help students understand the language of chemical equations. This worksheet is meant to be completed once students have learned the rules for chemical formulae, bonding, and state symbols. It also allows teachers to give feedback and suggestions on areas students might be struggling with. This worksheet consists of two parts: page one summarizes the rules of writing chemical equations. However, some of these rules will not be relevant for A Level students.
Balance chemical equations
A chemical equation can be broken down into two parts: a reactant or product. The equation is separated by an arrow, so in the example below, the reactants are H2 and O2 and the products are H20 and CO2. Balance a chemical equation requires that the products and reactants must be equal. It can be hard to balance a chemical formula. However, there are ways around this.
To help students learn how to balance chemical equations, there is a balancing chemical equations worksheet available online. The worksheet includes ten unbalanced equations as well as an answer key. It is a great resource for elementary students learning about the chemical process. The worksheet can be completed on a tablet by students. They can also check their understanding using the answer key.
When you are learning about balancing chemical equations, a balancing chemical equations worksheet is a great visual activity to help students understand the difference between subscripts and coefficients. The worksheet also explains why it is important to balance chemical equations, and how to do it. A balancing chemical equations worksheet allows you to easily create multiple worksheets. Each worksheet contains a matching answer sheet.
When you are learning about chemical equations, you have to understand the Law of Conservation of Mass. The Law of Conservation of Mass states that the amount of atoms on both sides of a chemical equation should be the same. First, find an element with a single reactant or one product to balance a chemical equation. Then, use this element to balance the first one.
Classify reactions
A chemistry equation describes a chemical reaction in mathematical terms. For example, magnesium reacts with oxygen gas to form a solid magnesium oxide, or calcium metal reacts with water to form calcium hydroxide precipitate. Ammonia is formed when nitrogen reacts with hydrogen gas to form gaseous ammonia. The reaction does not destroy ammonia, which is unlike other chemical reactions. Therefore, the atoms are unaltered.
Chemical reactions are a common part of everyday life. These processes change the chemical structure of reactants and produce new substances. These reactions cause changes in the bonds structures of substances. These changes can occur as a result of energy use or release. Many physical indicators can also be observed. These characteristics help classify reactions.
An acid-base reaction is a common chemical reaction in nature. Both reactants exchange electrons. When an acid reacts with a base, it neutralizes the acid. An acid and a base can also react to each other to create a new substance. Similarly, a precipitation reaction is a reaction in which the reactants disperse in a liquid. Depending on the solubility properties of the reactants, different precipitates can be formed. There are also redox reactions, which involve the transfer of electrons between two or more reactants and result in ionic products. In addition, hydrolysis reactions use water as one of the reactants. They produce smaller products such as CO and H2.
Determine coefficients
The first step in solving chemical equations is to determine the coefficients. The coefficients are whole numbers that represent the amount of each element in a chemical equation. These coefficients are calculated by balancing both the numbers of the elements on each side of an equation. If a chemical contains 10 atoms oxygen and 1 atom of sulfur, its coefficient for oxygen is 2. If the atoms on the opposite side of the equation have the same number, they are said to be a mixed compound.
To balance chemical equations, you should write the reactants and products correctly. You should also use symbols to indicate the state of substances. For example, if the substance is a salt, it should be written NaCl. It cannot be written as Na2Cl2.
The next step in solving chemistry equations is to find the balance between the atoms and molecules. We all know that the chemical equations must have equal amounts of both substances. Chemical equations must also be balanced, as each type of atom must be represented equally.
To determine the molar mass for a given element, you can use stoichiometric relationships to solve a chemical problem. By calculating the molar mass, you can convert Fe(s) into H2(g). The molar mass for H2O, for example, is twice that of a single H-atom.