Chemistry Endothermic Or Exothermic Reaction Worksheet Key Pdf – A Chemistry Reactions Worksheet is a useful tool to teach students the concepts of chemical change. A chemical reaction involves the transfer of energy between reactants and products. This type of change is either irreversible or reversible. It occurs when two atoms or molecules react to form a new product.
Changes in the bond structure can cause chemical reactions
Chemical reactions are processes that produce new molecules, usually by breaking or forming bonds between two substances. These reactions require energy because it takes energy to break bonds, and then release the product. Different types of bond structure produce different amounts of energy. A Lewis acid-base reaction, for example, produces a covalent bonds, in which the Lewis acid provides an electron pair, and the Lewis base receives one.
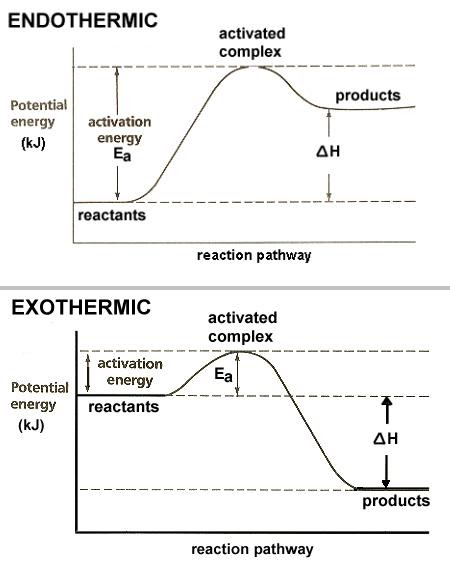
The energy involved in chemical reactions can be approximated using the bond strengths of reactants and products. The chemical reactions can cause these bond strengths to change. This energy can be measured in terms of heat, enthalpy and thermal energy. The energy of chemical reactions is also expressed at the atomic level as potential energy. This idea of energy is not often explained in chemistry textbooks.
These involve energy transfer between products and reactants.
In chemical reactions, energy is transferred from reactants to products. The energy is transferred through the form of bonds. This energy is called bond energy and is measured in kJ*mol$-1. The amount of energy that can be transferred in a chemical reaction depends on the amount of energy that the reactants and products have.
Understanding chemical reactions is key to understanding how energy is transferred. These reactions are characterized by energy change, i.e., energy absorption when chemical bonds break, or energy release when chemical bonds are formed. Generally, this energy is a form of heat or light, depending on the reactants and products. The energy transfer occurs because of the differences in stored chemical energy, or enthalpy.
They can be reversed
When both products and reactants are converted to one another in a chemical reaction, the process is known as a reversible reaction. It occurs when the conversion of the reactants to the products occurs simultaneously. This reaction is the most common in chemistry. This is how it works.
A reaction that occurs between a substance and a gas can be reversible or irreversible. A product is when an acid reacts to an alcohol. To allow this reaction to take place, it is necessary to let go of any gas molecules that were previously bound with the solution. A Dean-Stark apparatus is used to separate the reactants, ensuring that the desired product is produced.
They cannot be reversed.
Chemistry can produce many different types of reactions. Reactants and their surroundings will determine the type of reaction. The majority of chemical reactions can’t be reversed. They involve the conversion of two or more reactants into one or more products. Sometimes, the reaction is enhanced with the help of a catalyst.
A reversible reaction is one that occurs in a closed container. Ammonium chloride, for example, can be heated to make ammonia or hydrogen chloride. It is then converted to ammonium chloride when it cools. These two reactants will then recombine.
They involve redox reactions
Redox reactions involve the transfer of electrons between different chemical species. The oxidation process results in the loss of one or several electrons by an oxidizing agent, while the reduction process results in the gain of electrons from the reducing agent. Redox reactions can have a wide range of effects on environmental variables such as contaminant mobility or degradation. For example, hexavalent chromium is highly toxic when oxidized. In contrast, trivalent chromium is less toxic but less mobile. Likewise, arsenic, uranium, and selenium are less mobile under oxidizing conditions.
During decomposition, redox reactions may also occur. This results in a smaller chemical compound. For example, if CaCO3 reacts with CO2, it will decompose into CaO and CO2, but the oxidizing agent gains an electron. The oxidizing agent may also gain oxygen, bringing it into the molecule. Typical oxidative reactions in organic chemistry include dealkylation, epoxidation, aromatic ring cleavage, and hydroxylation.
They involve acids and bases
A Chemistry reaction involves acids and bases reacting with each other to produce a new substance. When the acid reacts with the base, it produces a new substance called a salt. Salts are crystal substances that dissolve in water. They are also bitter in taste. There are many theories as to how acids and bases interact with one another.
Acids and bases have important roles in chemical processes and are important in everyday life. The body’s acidity helps to maintain a stable internal environment. They also play an important role in baking a cake, and a lake’s acidity determines whether it can support aquatic life. A large number of chemical reactions involve either acids and bases. Acids and bases also play a key role in biological processes. The pH and alkalinity in the soil and water are crucial for animals and plants. The chemistry of acids or bases is a constant part of our everyday lives.